Not far at all.
The principal barriers to implementation are regulatory rather than technical. But prepare to be underwhelmed by the results.
Most of the action in applying the insights of microbiomics to obesity and related metabolic disorders has come in the form of probiotic (“good” bacteria) or prebiotic (compounds that favor the growth of “good” bacteria) interventions. Many of these have shown the hoped-for results in secondary measurements, such as micro biome composition, serum lipids, or insulin sensitivity. But most have failed to show meaningful differences in body mass or fat gain[1] [2] [3] [4] .
The research referred to in the question takes a different strategy – bacteria are engineered to synthesize a molecule (N-acylphosphatidylethanolamine, NAPE) that is a precursor to another molecule (N-acylethanolamide) which is synthesized in the upper small intestine and suppresses food intake and weight gain[5] .
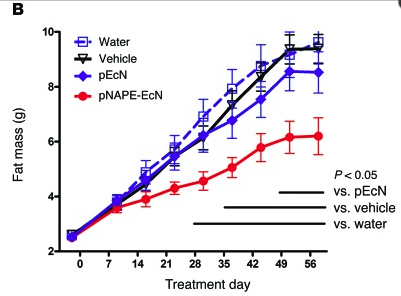
Davies’ group took a gene for synthesizing NAPE from a mustard weed and cloned it into the common gut bacterium E. coli. Mice fed on a high-fat diet and given the engineered bacteria gained much less fat than control mice[6] (red line):
The amount of bacteria given to the mice was enormous – 10^11 cells per day. Scaled up to a human, an equivalent dose would be about 100 grams of bacteria per day. That’s a lot of bugs to gag down, but they were only given for seven days, and the results appear to have persisted for much longer, once the engineered bacteria were able to establish themselves.
So far, so good. In principle, these engineered bugs might well be ready for clinical testing in humans. But it is not yet certain what regulatory hurdles this treatment would have to clear. The FDA is moving toward defining probiotics as a biological agent, requiring that they demonstrate safety and efficacy before therapeutic claims can be made[7] . Since the engineered bugs will inevitably enter the environment, the EPA will also have jurisdiction[8] . At this point, I don’t believe that there is an established path to market clearance.
The regulatory issues will be resolved eventually. The less-certain issue is whether this approach will actually work. Is giving a patient a bacterium that makes a therapeutic molecule really all that different than just giving them that same molecule? Anti-obesity drugs have a long history of working great in mice – which are genetically homogenous and whose diets and environments can be carefully controlled – and flopping in humans[9] [10] [11] . My guess is that any particular approach to microbiome engineering, much like targeted oncology drugs, will be successful only in some people some of the time. To be effective, these interventions will probably require both genotyping of the host, and an analysis of their existing microbiome.
Personalizing microbiome therapy in this way is completely feasible[12] , but will require a good deal more basic research – and probably some spectacular flame-outs of much-hyped companies and therapies – before it becomes truly effective.
Featured on Forbes
Footnotes
[1] Comparison of the effect of daily consumption of probiotic compared with low-fat conventional yogurt on weight loss in healthy obese women followin… – PubMed – NCBI
[2] Probiotic supplementation attenuates increases in body mass and fat mass during high-fat diet in healthy young adults.
[3] Dietary modulation of the gut microbiota–a randomised controlled trial in obese postmenopausal women.
[4] Randomised clinical trial: The beneficial effects of VSL#3 in obese children with non-alcoholic steatohepatitis.
[5] N-acylphosphatidylethanolamine, a gut- derived circulating factor induced by fat ingestion, inhibits food intake.
[6] Incorporation of therapeutically modified bacteria into gut microbiota inhibits obesity
[7] Federal Regulation of Probiotics: An Analysis of the Existing Regulatory Framework and Recommendations for Alternative Frameworks
[8] Restrictions on Genetically Modified Organisms: United States
[9] Drug treatment of obesity: from past failures to future successes?
[10] Leptin resistance – or why leptin fails to work in obesity.
[11] Leptin Fails Again: The Thin Line Between Hope and Hype
[12] Quantifying Diet-Induced Metabolic Changes of the Human Gut Microbiome.